Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (34): 5477-5483.doi: 10.3969/j.issn.2095-4344.0683
Previous Articles Next Articles
Bladder acellular matrix combined with vascular endothelial growth factor repairs rabbit urethral defects
Gu Shaodong1, Zhou Yun2, Xiong Qianwei2, Zhang Ya3, Yang Jinlong4
- 1Department of Pediatric Surgery, Lianyungang No. 1 Hospital, Lianyungang 222002, Jiangsu Province, China; 2Department of Pediatric Urology, 3Laboratory, Children Hospital of Soochow University, Suzhou 215025, Jiangsu Province, China; 4Department of Pediatric Surgery, Wuxi No. 8 People’s Hospital, Wuxi 214000, Jiangsu Province, China
-
Received:2018-07-10Online:2018-12-08Published:2018-12-08 -
Contact:Zhou Yun, Doctoral supervisor, Department of Pediatric Urology, Children Hospital of Soochow University, Suzhou 215025, Jiangsu Province, China -
About author:Gu Shaodong, Master, Associate chief physician, Department of Pediatric Surgery, Lianyungang No. 1 Hospital, Lianyungang 222002, Jiangsu Province, China -
Supported by:the Jiangsu Provincial Clinical Medical Science and Technology Special Project - New Clinical Diagnostic Technology Research Project, No. BL2012051
CLC Number:
Cite this article
Gu Shaodong, Zhou Yun, Xiong Qianwei, Zhang Ya, Yang Jinlong. Bladder acellular matrix combined with vascular endothelial growth factor repairs rabbit urethral defects[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(34): 5477-5483.
share this article
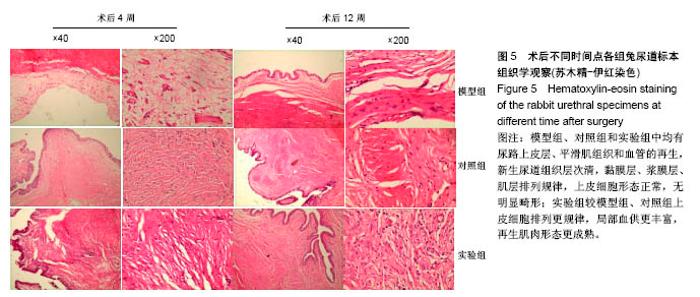
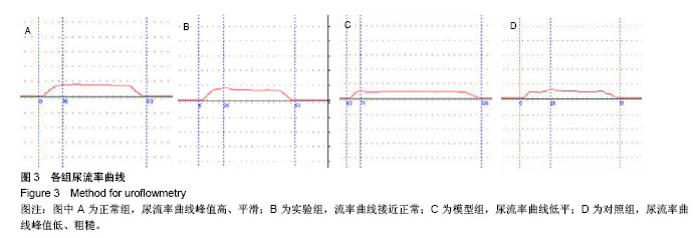
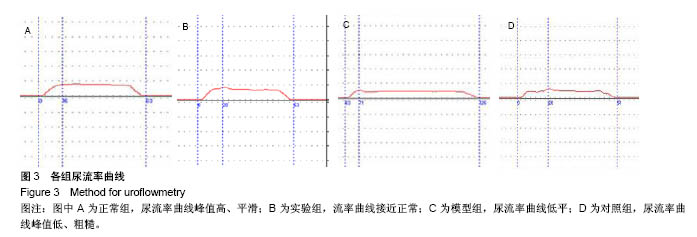
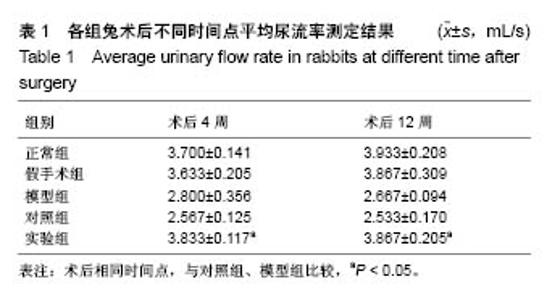
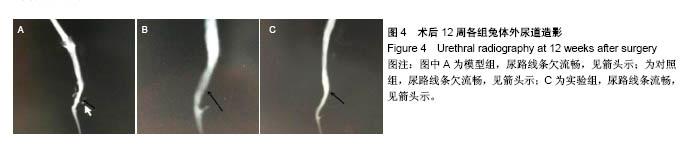
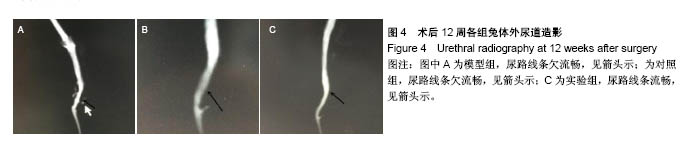
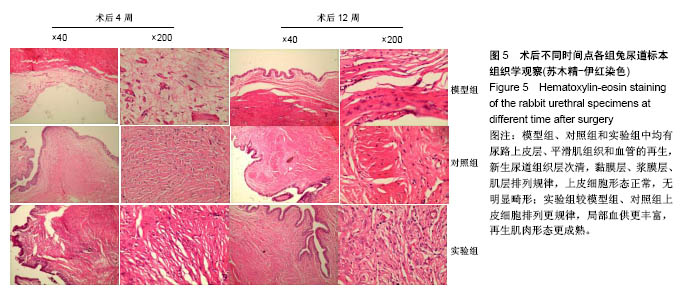
2.5 术后不同时间点各组尿道标本苏木精-伊红染色观察 模型组、对照组和实验组中均有尿路上皮层、平滑肌组织和血管的再生,新生尿道组织层次清,黏膜层、浆膜层、肌层排列规律,上皮细胞形态正常,无明显畸形。实验组较模型组、对照组上皮细胞排列更规律,局部血供更丰富,再生肌肉形态更成熟。 术后4周,模型组黏膜上皮厚度较周边薄;黏膜下有较多胶原纤维细胞生长,纤维排列紊乱,其间少量淋巴细胞浸润;对照组和实验组黏膜厚度基本正常,黏膜下胶原基质内有较多淋巴细胞浸润;实验组上皮修复良好,基质表面可见完整的黏膜覆盖,黏膜下层可见未完全降解的胶原基质,基质内有新的胶原纤维细胞生长,可见新生血管长入,对照组基质内未见明显新生血管。术后12周,模型组胶原纤维细胞进一步增多,纤维排列紊乱,有较多淋巴细胞浸润;实验组胶原基质基本降解,新生胶原纤维排列规律,其间血管较多,未见血管瘤等;对照组见团块状胶原沉积,视野内血管较少(图5)。"
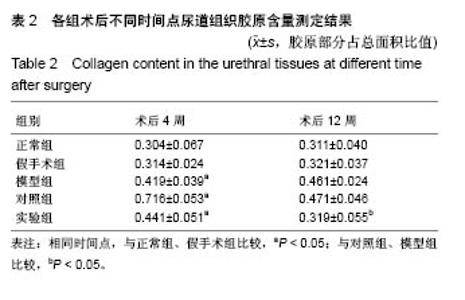
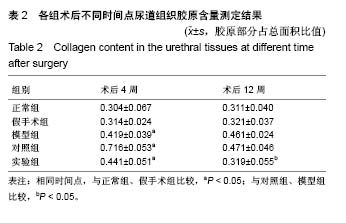
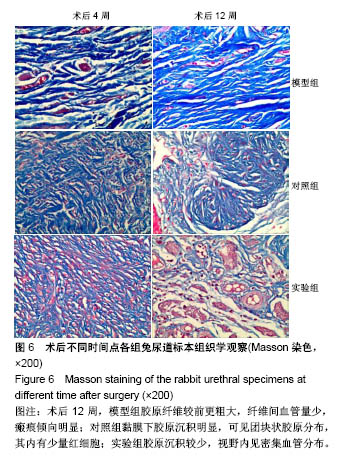
2.6 术后不同时间点各组尿道标本Masson染色胶原退化情况观察 术后4周,模型组黏膜下见粗大的胶原纤维,纤维间可见少量血管新生;实验组修补的移植物部分胶原含量较高,其内有较多红细胞分布;对照组胶原纤维粗大,纤维间红细胞数量明显较实验组少。术后12周,模型组胶原纤维较前更粗大,纤维间血管量少,瘢痕倾向明显;对照组黏膜下胶原沉积明显,可见团块状胶原分布,其内有少量红细胞;实验组胶原沉积较少,视野内见密集血管分布(图6)。 用IPP 6.0软件统计进行统计学处理,结果如下:术后4周,模型组、对照组、实验组胶原含量均高于正常组和假手术组(P < 0.05);术后12周,实验组胶原组织沉积明显少于对照组、模型组(P < 0.05),接近正常组和假手术组(P > 0.05),见表2。"
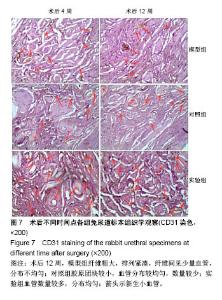
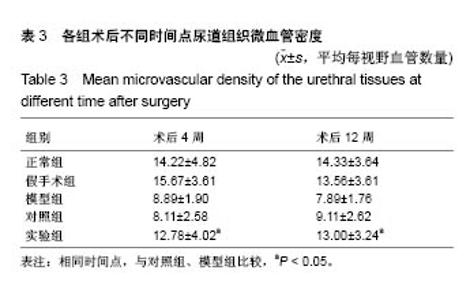
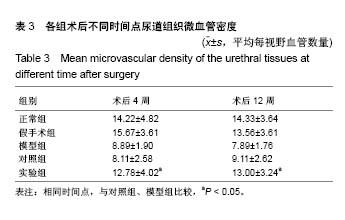
2.7 术后不同时间点各组尿道标本CD31染色新生血管情况观察 术后4周,模型组内纤维间少量血管分布,对照组胶原块周边可见少量血管分布,实验视野下见较多血管分布。术后12周,模型组纤维较4周时间更粗大,排列紧凑,纤维间见少量血管,分布不均匀;对照组胶原团块缩小,血管分布较均匀,数量较少;实验组血管数量较多,分布均匀(图7)。 由镜下观察发现,无论术后4周还是12周,不同组间新生血管的形态学均接近成熟,血管壁完整,血管通透性良好,无明显红细胞外溢,水肿等其他炎性表现。 经CD31染色剂染色后,用200倍视野下,每切片取3个以上视野对镜下的血管数目进行统计,观察统计各组尿道组织中血管数量,统计学分析可看出,同周期实验组明显优于对照组、模型组(P < 0.05),见表3。"
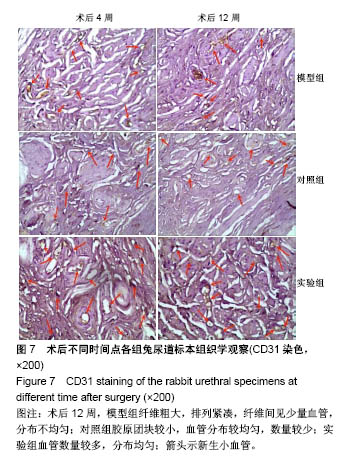
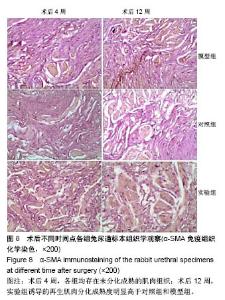
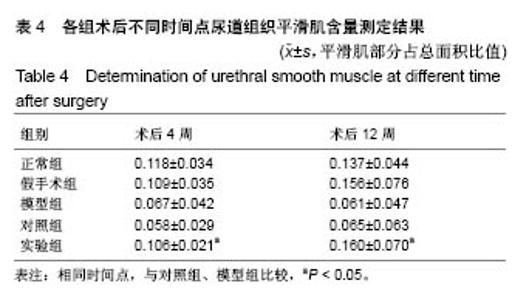
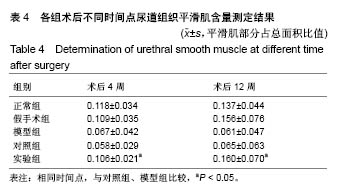
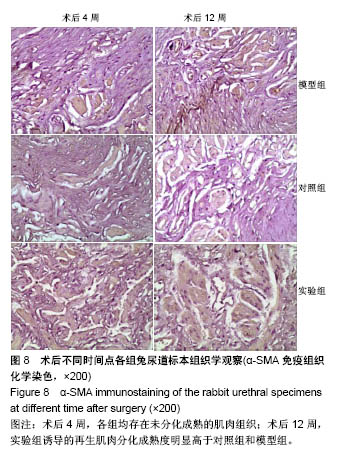
2.8 术后不同时间点各组尿道标本α-SMA免疫组织化学染色观察 α-SMA免疫组织化学染色后,肌肉呈淡黄色。术后4周,各组均存在未分化成熟的肌肉组织,表现为肌肉含量少,肌肉形态不规则,肌成纤维细胞假阳性率高等。术后12周,实验组诱导的再生肌肉分化成熟度明显高于对照组和模型组,表现为肌肉成规则状排列,形态结构完整,成条束状,肌肉组织层次清楚,考虑在VEGF诱导作用下,肌纤维细胞假阳性率降低,向肌肉细胞方向分化加强,肌肉含量较之前增多(图8)。 用IPP 6.0软件统计200倍放大视野内再生平滑肌所占面积百分比,可观察到,实验组再生肌肉含量明显高于对照组和模型组(P < 0.05),见表4。 "
| [1] 陈恕柱,靳宏勇,刘毅东,等.舌黏膜与口腔黏膜在治疗多次尿道下裂手术失败患儿中的临床应用和比较[J].中华小儿外科杂志, 2017,38(1):59-63. [2] 任晓敏,蒋跃庆,姚海军,等.后型尿道下裂二期膀胱黏膜半管状重建尿道术的临床研究[J].现代泌尿外科杂志, 2011,16(6):502-504. [3] Weiser AC,Franco I,Herz DB,et al.Single layered small intestinal submucosa in the repair of severe chordee and complicated hypospadias.J Urol.2003;(4 Pt.2):1593-1595; disussion 1595-0.[4] 李振武,张潍平,孙宁,等.国内医院尿道下裂治疗现状调查[J].中华小儿外科杂志,2016,37(6):453-458. [5] 肖元宏,刘贵麟,刘洲禄.Onlay岛状皮瓣尿道成形术治疗儿童尿道下裂适应证及并发症防治[J].解放军医学院学报, 2016,37(11):1152-1154,1159.[6] 梁伟强,冀晨阳,张佳琦,等.尿道口周蒂肉膜瓣在阴茎中段型尿道下裂一期修复中的应用[J].中国美容整形外科杂志, 2017,28(2):107-111. [7] 江志勇,李学德,何庆鑫,等.组织覆盖技术在Snodgrass术治疗尿道下裂的应用研究进展[J].中国性科学,2016,25(1):24-27.[8] 唐耘熳.尿道下裂术后尿道狭窄、阴茎头裂开及尿道憩室的认识及处理[J].临床小儿外科杂志,2017,16(3):212-214. [9] 王德娟,李科,黄文涛,等.游离舌黏膜在儿童再次尿道下裂成形术中的应用[J].新医学,2016,47(4):242-245. [10] Sievert KD,Amend B,Stenzl A.Tissue engineering for the lower urinary tract: a review of a state of the art approach.Eur Urol. 2007;52(6): 1580-1589.[11] Atala A.Recent developments in tissue engineering and regenerative medicine.Curr Opin Pediatr.2006;18(2):167-171.[12] Svystonyuk DA,Mewhort HEM,Fedak PWM.Using Acellular Bioactive Extracellular Matrix Scaffolds to Enhance Endogenous Cardiac Repair. Front Cardiovasc Med.2018;5:35[13] Udelsman B,Mathisen DJ,Ott HC.Bioprosthetics and repair of complex aerodigestive defects.Ann Cardiothorac Surg. 2018;7(2):284-292.[14] Emodi O,Ginini JG,van Aalst JA,et al.Cleft Palate Fistula Closure Utilizing Acellular Dermal Matrix.Plast Reconstr Surg Glob Open.2018; 6(3):e1682.[15] Griffith LG,Naughton G.Tissue engineering--current challenges and expanding opportunities. Science. 2002;295(5557):1009-1014. [16] Leslie B,Jesus LE,El-Hout Y,et al.Comparative histological and functional controlled analysis of tubularized incised plate urethroplasty with and without dorsal inlay graft: a preliminary experimental study in rabbits.J Urol.2011;186(4 Suppl):1631-1637.[17] Kanematsu A,Yamamoto S,Ogawa O.Changing concepts of bladder regeneration.Int J Urol. 2007;14(8):673-978.[18] Reyes M,Dudek A,Jahagirdar B,et al. Origin of endothelial progenitors in human postnatal bone marrow.J Clin Invest. 2002;109(3):337-346.[19] Nieda M,Nicol A,Denning-Kendall P,et al.Endothdial cell precursors are normal components of human umbilical cord blood.Br J Haematol. 1997;98(3):775-777.[20] Tamaki T,Akatsuka A,Ando K,et al.Identification of myogenic- endothelial progenitor cells in the interstitial spaces of skeletal muscle. J Cell Biol.2002;157(4):571-577.[21] Dahms SE,Piechota HJ,Dahiya R,et al.Composition and biomechanical properties of the bladder acellular matrix graft: comparative analysis in rat, pig and human.Br J Urol. 1998;82(3):411-419.[22] De Filippo RE,Yoo JJ,Atala A.Urethral replacement using cell seeded tubularized collagen matrices.J Urol.2002;168(4-2):1789-1792.[23] Shokeir A,Osman Y,El-Sherbiny M,et al.Comparison of Partial Urethral Replacement with Acellular Matrix versus Spontaneous Urethral Regeneration in a Canine Model.Eur Urol. 2003;44(5): 603-609. [24] Atala A,Guzman L,Retik AB.A novel inert collagen matrix for hypospadias repair.J Urol. 1999;162(3 Pt 2):1148-1151.[25] Kanematsu A,Yamamoto S,Noguchi T,et al.Bladder regeneration by bladder acellular matrix combined with sustained release of exogenous growth factor.J Urol. 2003;170(4 Pt 2):1633-1638.[26] Kanematsu A,Yamamoto S,Ozeki M,et al.Collagenous matrices as release carriers of exogenous growth factors. Biomaterials. 2004; 25(18):4513-4520.[27] Youssif M,Shiina H,Urakami S,et al.Effect of vascular endothelial growth factor Oil regeneration of blader acellular matrix graft:histologic and functional evaluation.Urology. 2005;66(1):201-207.[28] Wang JH,Xu YM,Fu Q,et al.Continued sustained release of VEGF by PLGA nanospheres modified BAMG stent for the anterior urethral reconstruction of rabbit.Asian Pac J Trop Med.2013;6(6):481-484.[29] Xiong Q,Lin H,Hua X,et al.A nanomedicine approach to effectively inhibit contracture during bladder acellularmatrix allograft-induced bladder regeneration by sustained delivery of vascular endothelial growth factor.Tissue Eng Part A. 2015;21(1-2):45-52.[30] Jiang X,Xiong Q,Xu G,et al.VEGF-Loaded Nanoparticle-Modified BAMAs Enhance Angiogenesis and Inhibit Graft Shrinkage in Tissue-Engineered Bladder.Ann Biomed Eng.2015;43(10):2577-2586.[31] Roelofs LA,Oosterwijk E,Kortmann BB,et al.Bladder Regeneration Using a Smart Acellular Collagen Scaffold with Growth Factors VEGF, FGF2 and HB-EGF.Tissue Eng Part A. 2016;22(1-2):83-92. [32] Wang L,Shi Q,Dai J,et al.vascularization promotes functional recovery in the transected spinal cord rats by implanted vascular endothelial growth factor-targeting collagenscaffold.J Orthop Res. 2018;36(3):1024-1034.[33] Ferrara N,Henzel WJ.Pituitary follicular cells secrete a novel heparin-binding growth factor specific for vascular endothelial cells.Biochem Biophys Res Commun.2012;425(3):540-547.[34] Koch S,Yao CH,Grieb G,et al.Enhancing angiogenesis in collagen matrices by covalent incorporation of VEGF.J Mater Sci Mater Med. 2006;17(8):735-741.[35] Yao N,Gao M,Ren K,et al.PD806: a novel oral vascular disrupting agent shows antitumor and antivascular effects in vitro and in vivo. Anticancer Drugs.2015;26(2):148-159.[36] Giatromanolaki A,Koukourakis MI,Theodossiou D,et al. Comparative evaluation of angiogenesis assessment with anti-factor-VIII and anti-CD31 immunostaining in non-small cell lung cancer. Clin Cancer Res.1997;3(12 Pt 1):2485-2492. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||